Which of the Following Is the Best Leaving Group
Typically the leaving group is an anion eg. O II 10 O V O IV.

Regiochemistry Of Hofmann Elimination The Steric Effect Chemistry Lessons Chemistry Classroom Chemistry
Experts are tested by Chegg as specialists in their subject area.

. A F- B CI- C I- D Br- E H2N-. Place in order of decreasing leaving group ability best leaving group first. The best leaving group is the atomion that will be the most stable species when it is kicked out of the substrate.
Hence option E is correct. Rank the following ion in terms of their ability as potential leaving groups remember the more stable more weakly basic the leaving group the better it is. Now In the all above examples Bro is the best leaving group because Bre atom inabizes has big size thats why it attached to the Carbon not very strongly so it can caisly break the bond to the carbon whereas.
They dont want to share them with other atoms. I- Br- H2N- HO-. NH2 I F H2O OCH3 C6H5SO3 OH NH3.
Chlorine is a good leaving group and fluorine is a poor leaving group. Which of the following is the best leaving group. Place the worst leaving group at the top of the list.
Good leaving groups are weak bases. The best leaving groups want those electrons. Which of the following statements about the SN1 mechanism is correct.
All are equally good leaving groups. A leaving group LG is an atom or a group of atoms that is displaced as stable species taking with it the bonding electrons. Consider a general nucleophilic substitution reaction.
Iodine is the best leaving group due to the larger size of iodine. It acts as best nucleophile because it is able to donate lone pair of electrons easily. Which of the following is the best leaving group.
The better the leaving group the more likely it is to depart. HO HO B SN C E D E A SN E Eicb HS substitutes Br from. The SN1 mechanism involves a single reaction step.
The mechanism has only one step. C Br E They are all equal A F DI B Cl In the following reaction when the concentration of OH was doubled keeping the concentration of 1-idodopropane the same the rate of the reaction was doubled. I need the end answer quickly.
Size of F is to small so it bonded to the carbon atom too lightly. What is the reaction mechanism. Best Leaving Group in Substitution.
Who are the experts. In this case -NH 2 will be considered as the worst among the group since the other LGs are smaller. All halide anions except F- are good leaving groups because of their conjugate acids HCl HBr and HI have low pKa.
Explain your answer briefly. The stereochemical outcome is racemization at the carbon bearing the leaving group. The leaving group dissociates leaves leaving behind a free radical d.
Cl - or a neutral molecule eg. The halogens iodine and bromine are both very good leaving groups. Weak bases have strong conjugate acids.
The second arrow always shows a pair of electrons going toward the leaving group. 人 Cı HO HN2 II II IV V Select one. The leaving group dissociates leaves leaving behind a carbocation c.
Best leaving group are self detachable. We use factors affecting acidity to determine which leaving groups will be most stable after gaining a lone pair. The leaving group is ʻpushed offʼ by the incoming nucleophile b.
H - Br conjugate base Generally good leaving groups are weak bases. So it is not good leaving group. N 2 is self detachable.
Which is the best leaving group in a substitution reaction of an alkyl halide from the following choices. Which of the following is the best leaving group. We review their content and use your feedback to keep the quality high.
Which of the following statements is not true regarding SN1 reactions. Rank the following compounds in order of increasing reactivity in nucleophilic acyl substitution reactions starting with the least reactive compound. Polar protic solvents are good choices for SN1 reactions.
A carbocation intermediate is formed. Asked Aug 25 2019 in Chemistry by Lucys. N 2 is best leaving group among given.
Good leaving groups are weak bases. Recall that the element effect consists of two trends.

Zaitsev S Rule Regioselectivity Of E2 Elimination Reactions Practice Problems Chemistry Lessons Reactions Organic Synthesis

Pin On Reactions Of Carboxylic Acids And Their Derivatives Practice Problems

Alcohol Reaction With Hcl Hbr And Hi Acids Chemistry Steps Chemistry Organic Molecules Organic Chemistry

Draw The Mechanism And Show The Products With Correct Stereochemistry For The Following Sn2 Reactio Chemistry Lessons Organic Chemistry Study Organic Chemistry
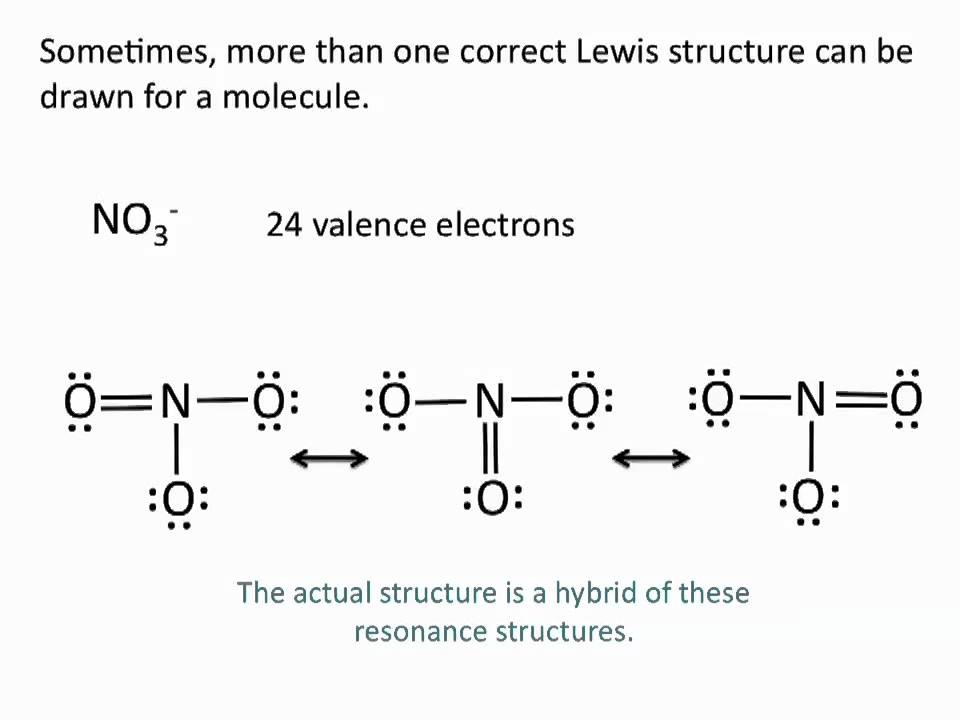
Drawing Lewis Structures Resonance Structures Chemistry Tutorial Youtube Chemistry Science Education Organic Chemistry

Sn1 And Sn2 Summary Cheat Sheets Chemistry Lessons Chemistry Study Guide Organic Chemistry

Leaving Groups Chemwiki Organic Chemistry Chemistry Organic Chem

Nucleophilic Substitution Reactions An Introduction Organic Chemistry Reactions Chemistry Reactions

The E1 And Sn1 Reactions Always Compete Since Both Form The Same Carbocation Organic Chemistry Reactions Chemistry

What Makes A Good Leaving Group In Nucleophilic Substitution And Elimination Reactions Chemistry Help Chemistry Teaching Chemistry

Zaitsev And Hofmann Products Regioselectivity Of E2 Elimination Reactions Organic Chemistry Chemistry Organic Synthesis

Worksheet Polarity Of Bonds Answers Worksheet Polarity Bonds Answers 221 Best Chemistry Chemistry Classroom Teaching Chemistry Chemistry Lessons

The Relative Rate Of E2 Reaction For Substituted Cyclohexanes Organic Chemistry Study Organic Chemistry Study Chemistry

What Makes A Good Leaving Group In Nucleophilic Substitution And Elimination Reactions Chemistry Help Chemistry Teaching Chemistry

Draw The Mechanism And Show The Products For The Following Sn2 Reactions Paint Program Online Painting Painting Teacher

Organic Chemistry Infographic Leaving Groups Organic Chemistry Science Doodles Chemistry



Comments
Post a Comment